Conjugate Acids and Conjugate Bases:
1. Conjugate acids and conjugate bases come from the Bronsted-Lowry Theory which states:
I know it's still a little bit confusing, so let's look at these examples to clarify everything:
Example #1-
1a. First, let's figure out what is the proton donor and what is the proton acceptor.
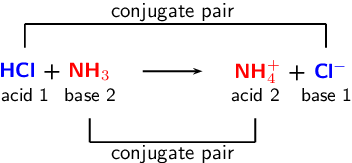
*In this reaction, our acid (HCl) is a monoprotic acid because it has one H+ ion.
HCl + NH3 -> NH4+ + Cl-
Acid/ Base/ + because it - because it
Proton Donor Proton Acceptor gains an H+ ion loses an H+ ion
*The color represents where the H+ ion is being displaced to.
1b. Now, let's use that same example to show which is the conjugate acid and which is the conjugate base:
HCl + NH3 -> NH4+ + Cl-
Acid Base Conjugate Acid Conjugate Base
*The [H+] molecule from the HCl (acid) is 'donating' itself to NH3 (base). Cl- is the conjugate base because it looses an H+ molecule, and NH4+ is the conjugate acid because it is 'accepting' the H+ that was 'donated' by the HCl.
Here is a picture of example #1-
- All acids are proton H+ donors, and all bases are proton H+ acceptors.
I know it's still a little bit confusing, so let's look at these examples to clarify everything:
Example #1-
1a. First, let's figure out what is the proton donor and what is the proton acceptor.
*In this reaction, our acid (HCl) is a monoprotic acid because it has one H+ ion.
HCl + NH3 -> NH4+ + Cl-
Acid/ Base/ + because it - because it
Proton Donor Proton Acceptor gains an H+ ion loses an H+ ion
*The color represents where the H+ ion is being displaced to.
1b. Now, let's use that same example to show which is the conjugate acid and which is the conjugate base:
HCl + NH3 -> NH4+ + Cl-
Acid Base Conjugate Acid Conjugate Base
*The [H+] molecule from the HCl (acid) is 'donating' itself to NH3 (base). Cl- is the conjugate base because it looses an H+ molecule, and NH4+ is the conjugate acid because it is 'accepting' the H+ that was 'donated' by the HCl.
Here is a picture of example #1-
Hopefully we're understanding this topic a little bit better, so let's do another example:
Example #2-
2a. Just like the first example, let's figure out which one is the proton acceptor and which one is the proton donor.
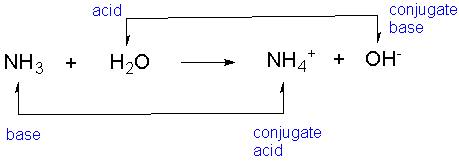
*In this reaction, our acid (H2O) is a diuretic acid because it has two H+ ions.
H2O + NH3 -> NH4+ + OH-
Acid/ Base/ + because it - because it
Proton Donor Proton Acceptor gains an H+ ion loses an H+ ion
*Before we move on I want to answer a question that I know you have:
How is H2O the acid/proton donor if H2O has a pH of 7 and is neutral (neither an acid or base)?
Although that is true, H2O is a proton donor, classifying it as an acid under the Bronsted-Lowry Theory. When it 'donates' a H+ ion, it becomes a conjugate base because it is giving away a H+ ion.
2b. Now that we know which is our proton donor and which is our proton acceptor, let's select our conjugate acid and our conjugate base:
H2O + NH3 -> NH4+ + OH-
Acid Base Conjugate Acid Conjugate Base
Here is a picture of example #2:
Example #2-
2a. Just like the first example, let's figure out which one is the proton acceptor and which one is the proton donor.
*In this reaction, our acid (H2O) is a diuretic acid because it has two H+ ions.
H2O + NH3 -> NH4+ + OH-
Acid/ Base/ + because it - because it
Proton Donor Proton Acceptor gains an H+ ion loses an H+ ion
*Before we move on I want to answer a question that I know you have:
How is H2O the acid/proton donor if H2O has a pH of 7 and is neutral (neither an acid or base)?
Although that is true, H2O is a proton donor, classifying it as an acid under the Bronsted-Lowry Theory. When it 'donates' a H+ ion, it becomes a conjugate base because it is giving away a H+ ion.
2b. Now that we know which is our proton donor and which is our proton acceptor, let's select our conjugate acid and our conjugate base:
H2O + NH3 -> NH4+ + OH-
Acid Base Conjugate Acid Conjugate Base
Here is a picture of example #2: